This page is currently under construction
Why choose Mobi-C® Cervical Disc over Fusion?
- Mobi-C the first and only cervical disc in the U.S. approved to treat more than one level of the cervical spine. It received FDA approval for one and two-level indications in August 2013.
Mobi-C demonstrated statistical superiority compared to cervical spine fusion in overall study success when used in two-level patients.
- Mobi-C has been implanted more than 17,000 times in 25 countries around the world since 2004.
- Mobi-C is differentiated from other cervical discs because of its bone sparing technique, which eliminates the need for bone chiseling and drilling, and optimizes it for two-level indications.
- Patented Mobile Core Technology™ allows the Mobi-C disc to angulate and slide in multiple directions similar to natural cervical motion.
- The greatest clinical benefit of artificial disc replacement with Mobi-C compared to cervical spine fusion were seen in two-level indications, particularly in reoperation rates and disability improvement.
- In the Mobi-C clinical study, patients returned to work on average one week faster (one-level patients) and three weeks faster (two-level patients) compared to fusion patients.
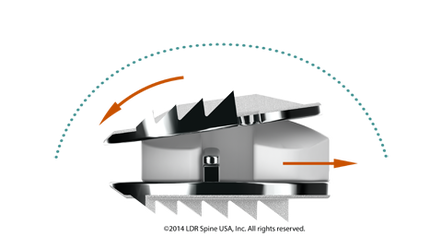
Mobi-C How it Works
Mobi-C contains patented mobile bearing technology that allows the polyethylene core to slide and rotate inside the disc for self-adjustment to the cervical spine movements. This means that Mobi-C can react to the normal motion in the cervical spine.
Mobi-C contains patented mobile bearing technology that allows the polyethylene core to slide and rotate inside the disc for self-adjustment to the cervical spine movements. This means that Mobi-C can react to the normal motion in the cervical spine.
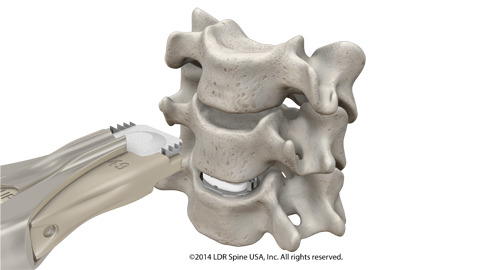
In a surgery with the Mobi-C Cervical Disc, the unhealthy disc is removed, but instead of a bone spacer or plastic implant along with a plate and screws, a Mobi-C is implanted into the disc space. Where a fusion procedure is intended to eliminate motion at the surgery levels, the goal of a surgery with Mobi-C is to allow motion at those levels.
Mobi-C provides bone sparing fixation without chisel cuts into the small vertebral bodies of the cervical spine, making it bone sparing.
As with any surgery, complications may occur as a result of treatment. Potential complications following treatment with the Mobi-C include:
- Pain in the neck, arm, back, shoulder, or head.
- The feeling of pins and needles in the arms.
- Difficulty swallowing.
Please visit www.cervicaldisc.com/aboutyourneck for a complete list of potential risks and benefits of Mobi-C surgery. Visit www.cervicaldisc.com/clinical-results for indications and detailed clinical results.
